
Revisiting how the immune system works to make better drugs
In the first of our new series entitled ‘Study in Focus’ Dr Doug Dyer writes about recent work from his lab led by Dr Anna Gray on the importance of chemokines in recruiting immune cells into tissues to fight infections and how a greater understanding of this process could lead to treating disease in a revolutionary way.
The problem
We have recently published a study focused on better understanding how the immune system works in order to be able to try and treat diseases in new ways. Our body’s immune system is made up of different cells that fight infection. A number of immune cells are made in the bone marrow and must travel via blood vessels to the places where they are needed.
Therefore a central question in the immune system is how cells are recruited to sites of infection in a tissue, e.g. lungs, from the circulation. Crucially this process is central to a wide range of inflammatory-based diseases such as COVID-19, rheumatoid arthritis, sepsis, and also cancer. Despite the importance of this recruitment process we still do not fully understand how it works.
Whilst the recruitment of immune cells from circulation into tissue is complex, the involvement of one family of proteins, chemokines, is known to be particularly important. My lab is investigating how chemokines function with the ultimate aim of treating disease in a new way. Our recent work has suggested that one chemokine CXCL4 (PF4) allows immune cells to reach tissues in a different way than other chemokines that have not previously been understood. In particular, we show that the building blocks of a tissue, the extracellular matrix, plays a more central role in the function of chemokines than was previously understood.
As discussed in a recent review the extracellular matrix is a critical, but under-explored component of the immune system. We hope this reveals a new opportunity to make drugs to treat diseases such as COVID-19, sepsis, and cancer.
The backstory
The story started nearly ten years ago when I was working at UCSD in the lab of Professor Tracy Handel. I was doing an experiment for a friend, Dr Catherina Salanga, to help her finish her research paper on the chemokine CCL2. She had asked me to work out how tightly her chemokine CCL2 bound to a purified extracellular matrix sugar component, the glycosaminoglycan heparan sulfate (HS).
The first experiment went well but when I went to repeat it the result looked completely different and as a new postdoc I was worried I had made a mistake. Crucially Dr Salanga and Prof. Handel encouraged me to look past the mistake and think about why the result looked so different. Thanks to their advice, the difference between the experiments turned out to be how I had prepared the matrix GAG sugar surface I was using to examine chemokine binding.
This led to a series of papers where we demonstrated that some chemokines could cross-link matrix GAG sugar chains. In collaboration with Dr Ralf Richter and following a visit to his lab in San Sebastian, Spain, we demonstrated that the chemokine CXCL4 was unique in its ability to bind and cross-link these extracellular matrix chains. For a variety of reasons I was really excited about all these findings but this still left us with the question of what this would mean to “real-world” biology and the immune response.
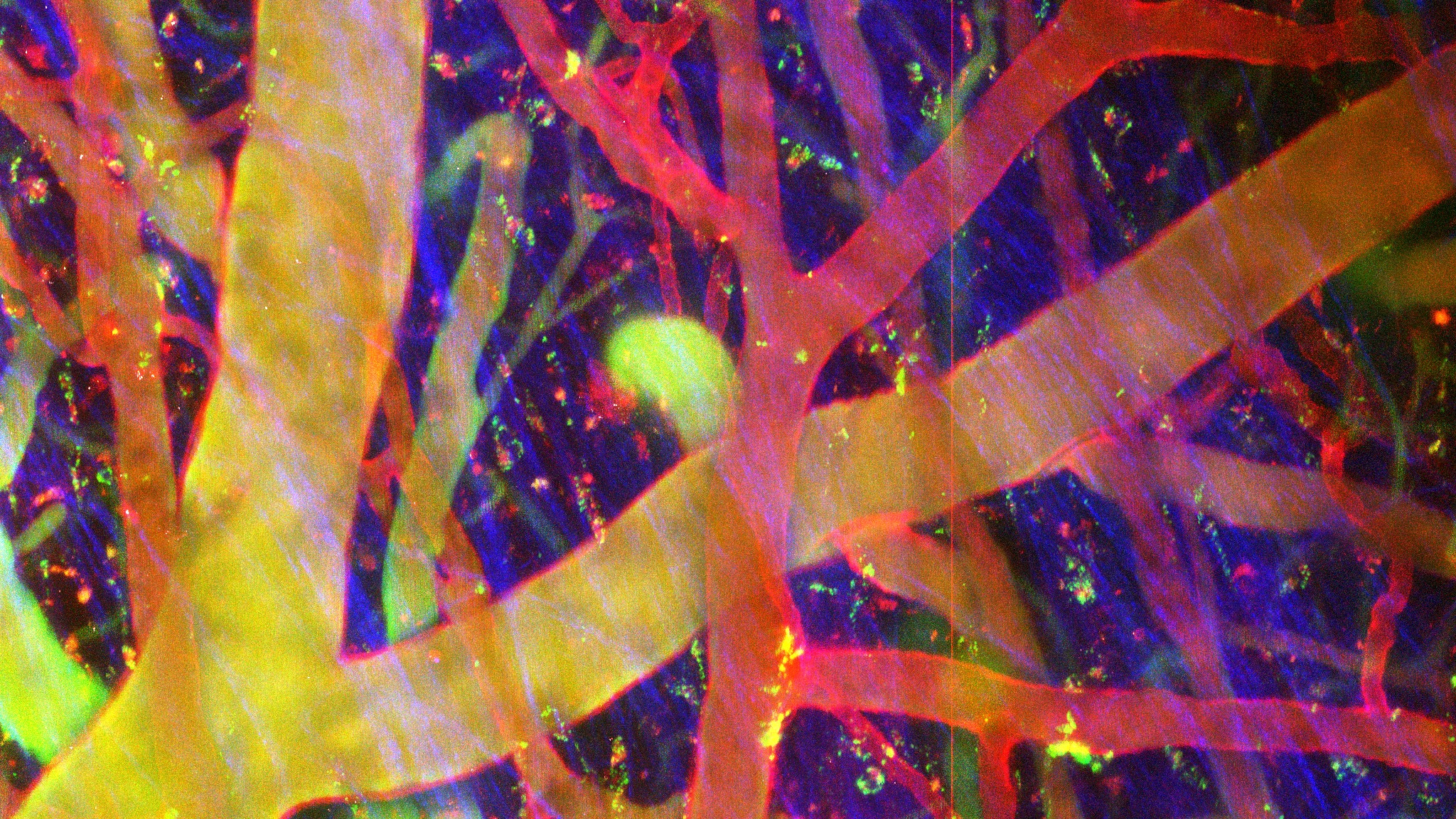
Image: Dr Anna Gray and Dr Ingo Schiessl
Our research
After working with Prof. Gerry Graham in Glasgow, Scotland I then relocated to Manchester to start my own lab and continued to work on my “mistake” on CXCL4. Working with postdocs Dr Anna Gray and Dr Amanda Ridley we applied biological skills to firstly show that in mice CXCL4 can recruit a wide range of immune cells during inflammation, this is very unusual as most chemokines only recruit one or two different types of immune cell.
In order to understand how this might be happening we then used human cells as well as mice to show that CXCL4 was in fact acting on endothelial cells lining the blood vessels, promoting their ability to facilitate the movement of immune cells from the circulation and into tissues. This was built upon by a masters student, Bakhtbilland Khan, who used an exciting mass-spectrometry approach, in collaboration with our core facility, to show that CXCL4 was actually signaling through the endothelial cells to allow them to facilitate immune cell migration. However, the receptor enabling binding of CXCL4 to endothelial cells remained unknown.
Our next task then was to discover what this receptor was! To do this we worked with the Manchester BioMols core facility and our collaborators in Copenhagen (Dr Rebecca Miller) to show that CXCL4 was binding to extracellular matrix GAGs on endothelial cells. We again collaborated with Dr Richter to show that this was due to the ability of CXCL4 to cross-link GAG chains, confirming the importance of our accidental observation almost ten years ago! Excitingly we also showed that an antibody aimed at inhibiting crosslinking of CXCL4 completely reversed its’ ability to recruit immune cells in mice.
The future
Now we need to exploit our new knowledge to develop new drugs to treat the wide range of diseases that CXCL4, and chemokines more generally, regulate. In particular, we are excited to further reveal the ways that the extracellular matrix can regulate the immune response providing opportunities for a new class of drugs.

Dr Doug Dyer – Wellcome Trust Sir Henry Dale Fellow, Lydia Becker Institute of Immunology and Inflammation, University of Manchester





0 Comments